URIC ACID, Determining uric acid level by The Caraway method and kit systems
URIC ACID
Uric acid is
the final breakdown product of purine metabolism. Purines such as adenosine and
guanine result from the breakdown of nucleic acids.
 They are
either ingested or come from the destruction of tissue cells, and are converted
mainly in the liver into uric acid. From the liver, uric acid is transported to
the kidneys where it is filtered by the glomeruli.
They are
either ingested or come from the destruction of tissue cells, and are converted
mainly in the liver into uric acid. From the liver, uric acid is transported to
the kidneys where it is filtered by the glomeruli.
Nearly all
the filtered uric acid is reabsorbed in the proximal tubules; and small amounts
are then secreted by the distal tubules and ultimately appear in the urine.
Nearly all
of the uric acid in the plasma is in the form of monosodium urate. Urates in
this form are relatively insoluble. At high levels (> 6.4 mg/dl ), the
plasma is saturated; urate crystals may form and precipitate in the tissues.
Assay Methods for Serum/Plasma Uric
Acid
Uric acid is
readily oxidised to allantoin and so can function as a reducing agent in many
chemical reactions. The Caraway method
is the most popular method for determining uric acid level. This method is
based on the oxidation of uric acid in a protein-free solution, with the
eventual reduction of phosphotungstic acid to tungsten blue. Sodium carbonate
is used to provide the alkaline pH necessary for colour development.
Unfortunately, this method is relatively non-specific, and so several methods
that make use of the specific enzyme, uricase, have been developed. This enzyme
catalyses the oxidation of uric acid to allantoin with the subsequent
production of hydrogen peroxide (H2O2). The uricase method is based on the fact
that uric acid has a significant UV absorbance with a peak at 293 nm, whereas
allantoin does not have. Therefore, the difference in absorbance in an
ultraviolet spectrophotometer before and after incubation with uricase is
proportional to the concentration of uric acid.
Caraway Method
Reagents
(i) Phosphotungstic acid reagent Dissolve 40.0 g of high grade sodium
tungstate in about 300 ml of distilled water. Add 32 ml of orthophosphoric acid
(H2PO4, 85% w/v). Reflux for 4 hours under a vertical condenser. Cool to room
temperature, and add about 300 ml of distilled water. In this solution,
dissolve 32.0 g lithium sulphate monohydrate and make volume up to 1 litre. Add
81.0 ml of 2 M sodium hydroxide solution and mix. The final pH is adjusted to
2.5; and the colour should be pale greenish-yellow.
(ii) Sodium carbonate solution Dissolve 140 g anhydrous sodium
carbonate in, and make up to 1 litre with distilled water. Mix well and store
in a plastic bottle.
(iii) Uric acid standard (stock)
50mg/dl Add 1.0 g of
lithium carbonate to about 500 ml distilled water. Warm to 60°C to dissolve.
Place 500 mg of pure uric acid in a 1 litre volumetric flask and add the
lithium carbonate solution to it. Warm to 60°C to dissolve. Cool and add 5.0 ml
formalin.
Add 400 ml
distilled water and 10 ml of 50 % acetic acid. Dilute to 1 litre with distilled
water.
(iv) Uric acid standard, working
(5mg/dl) Dilute the
stock standard 1:10 by diluting 10 ml to 100 ml with distilled water.
Technique
Prepare a
set of 3 tubes as follows:
test
|
Standard
|
Blank
|
|
Test Phosphotungstic acid reagent
|
2 ml
|
2 ml
|
2 ml
|
Sample 0.2 ml
|
0.2 ml
|
||
Working standard uric acid (5 mg/dl) -
|
0.2 ml
|
||
Distilled water
|
ml
|
(I)Add the
reagents as shown above
(ii) Mix
well and leave at room temperature for 15 minutes.
(iii)
Centrifuge all tubes at 3000 rpm for 10 minutes.
(iv) Decant
the supernatant directly into 3 separate correspondingly labelled tubes.
(v) Add 1.0
ml of sodium carbonate solution to each tube.
(vi) Leave
at room temperature for 15 minutes.
(vii) Read
the absorbance at 650-700 nm, setting the zero with the blank.
Kit systems Nearly all the kit systems are based
on the enzyme uricase method. They are specific, and the manufacturer's leaflet
clearly states in simple language, the method of assay.
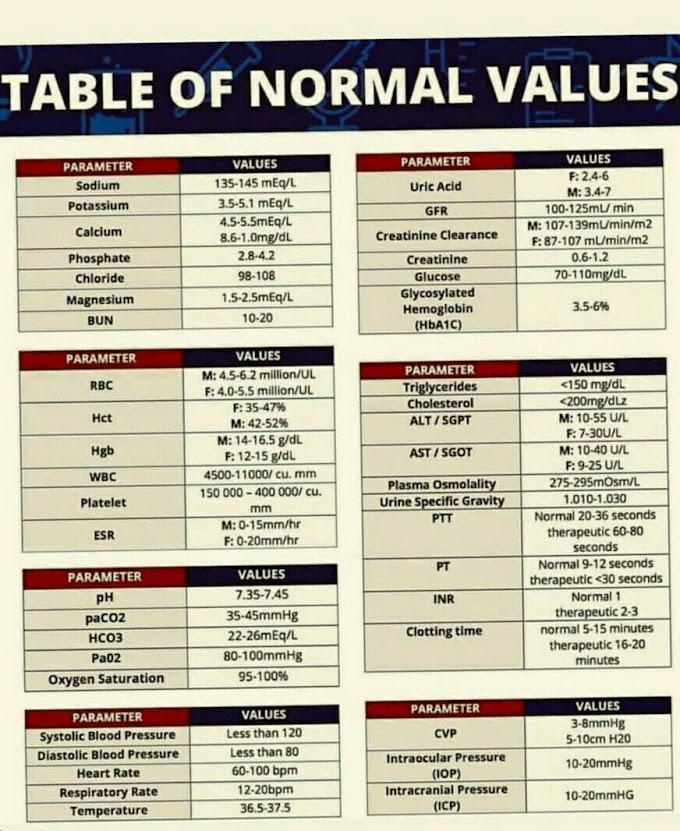
Reference Ranges for Uric Acid
Children:
2-5.5 mg/dl (0.12-0.32 mmol/L)
Adult males:
3.5-7.2 mg/dl (0.21-0.42 mmol/L)
Adult
females: 2.6-6.0 mg/dl (0.15-0.35mmol/L)
Interpretation
Factors
affecting uric acid levels in the blood Increased production, raised serum
levels (hyperuricaemia)
(i)
Idiopathic
primary gout
(ii)
Excessive
dietary purines (liver, kidney, legumes, etc.)
(iii)
Cytotoxic
treatment of malignancies such as leukaemias and lymphomas
(iv)
Polycythaemia
(v)
Multiple
myeloma
(vi) Sickle cell anaemia.
Decreased excretion, raised serum
levels
|
Decreased level of uric acid
(hypouricaemia) It is not very common but can be caused by :
|
(i)
Alcohol
ingestion
|
(i) Over treatment with allopurinol
|
(ii) Lactic acidosis, Ketoacidosis
|
(ii) Ingestion of drugs like aspirin
with doses above 4 g a day
|
(iv) Thiazid diuretics
|
(iii) Corticosteroids
|
(V) Renal dysfunction
|
(iv) Estrogens
|






If you have any queries related medical laboratory science & you are looking for any topic which you have have not found here.. you can comment below... and feedback us if you like over work & Theory
.
Thanks for coming here..