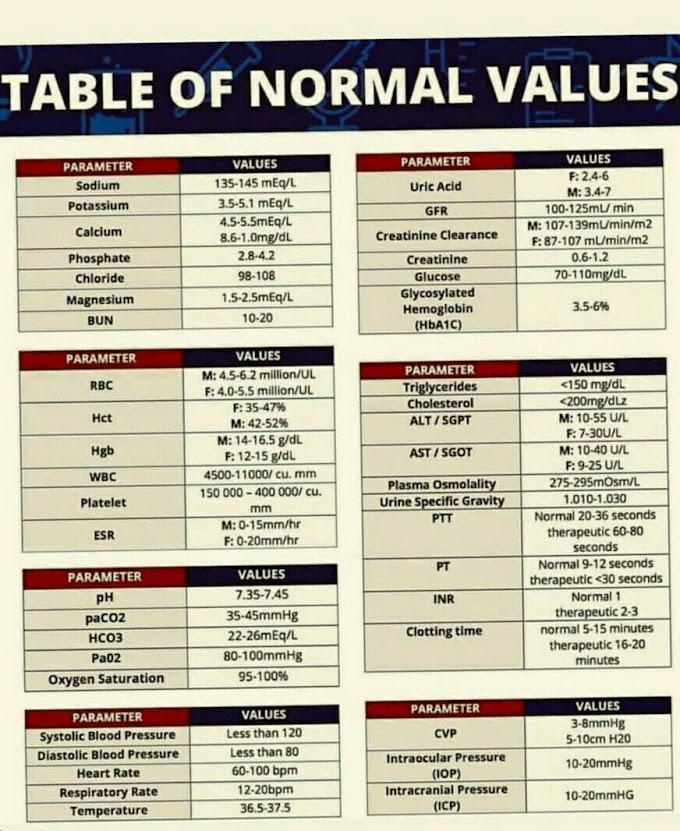
SPECTROSCOPIC
EXAMINATION OF BLOOD FOR THE DETECTION OF ABNORMAL HAEMOGLOBIN PIGMENTS
Certain
chemicals and drugs may react with haemoglobin to form abnormal pigments which
affect its capacity to bring about gaseous exchange.
Three such pigments,methaemoglobin,sulphhaemoglobin and carboxyhemoglobin,can be differentiated from each other
with the help of a spectroscope.
The
spectroscope splits white light into its component colours with the help of a
prism or a diffraction grating. Some substances absorb light at various
wavelengths.
When viewed through the eyepiece of the spectroscope, this
absorbed light appears as a dark band at that particular wavelength. Such
absorption bands are specific for each compound and is called an absorption
spectrum of that substance.
 Technique
Dilute the sample of blood 1:10 or 1:20 with distilled water and view it
through a spectroscope. Absorption bands appear either in the green or the red
part of the spectrum, depending on the type of the pigment present (Fig. 6.5)
Technique
Dilute the sample of blood 1:10 or 1:20 with distilled water and view it
through a spectroscope. Absorption bands appear either in the green or the red
part of the spectrum, depending on the type of the pigment present (Fig. 6.5)
Oxyhaemoglobin
(normal Hb) shows a narrow band at 575 nm (yellow region) and a wider band at
540 nm (green region).
Methaemoglobin
shows three bands which may not be very well defined, at 630 nm, 575 nm and 538
nm.
Sulphhaemoglobin
also shows three absorption bands at 618 nm, 575 nm and 538 nm.
Carboxyhaemoglobin
has an absorption spectrum very similar to that of oxyhaemoglobin, with bands
at 570 nm, 534 nm and a weak band at 418 nm.
Note
Simple
chemical tests help in the differentiation of hemoglobin pigments if the
absorption bands are not very clear. For example,
Dilute the
blood sample 1:500.
The normal oxyhaemoglobin will show a yellowish-red color. If, however, carboxyhaemoglobin is present in it, a purplish tint will be observed.
The normal oxyhaemoglobin will show a yellowish-red color. If, however, carboxyhaemoglobin is present in it, a purplish tint will be observed.
To one drop
of blood on a slide, add two drops of sodium hydroxide (NaOH, 250g/L) solution.
A normal specimen of blood will change to a
brownish red colour whereas that containing carboxyhaemoglobin will remain red.
iii. The
absorption bands of methaemoglobin and sulphhaemo-globin are very similar.
To differentiate between them, add 2-3 drops of sodium cyanide solution (50g/L) to a solution of haemoglobin. The cyanide will convert methaemoglobin into cyanmethaemoglobin which does not show any absorption band at the red region (630 nm).
Absorption spectrum of sulphaemoglobin remains unchanged.
To differentiate between them, add 2-3 drops of sodium cyanide solution (50g/L) to a solution of haemoglobin. The cyanide will convert methaemoglobin into cyanmethaemoglobin which does not show any absorption band at the red region (630 nm).
Absorption spectrum of sulphaemoglobin remains unchanged.






If you have any queries related medical laboratory science & you are looking for any topic which you have have not found here.. you can comment below... and feedback us if you like over work & Theory
.
Thanks for coming here..